Table of Contents
- The Supreme Court Preserved Access For Now
- Mifepristone Is At The Center Of The Fight
- Louisiana Challenged The FDA Rule
- Doctors Warned Confusion Harms Patients
- Abortion Opponents Condemned The Decision
- The Comstock Act Reentered The Debate
- Alito Focused On Irreparable Harm
- The FDA Safety Review Adds Another Layer
- Providers Are Preparing Backup Plans
- Telehealth Became A Critical Access Point
- Doctors Warn Travel Barriers Could Increase
- The Case Could Shape Abortion Access Nationwide
- The Political Battle Is Only Growing
- What Happens Next
The Supreme Court Preserved Access For Now
The Supreme Court’s Thursday order allowed telehealth access to mifepristone to continue while the 5th Circuit Court of Appeals considers the broader legal challenge.
That means patients can still access the medication under current FDA rules, including through telehealth where permitted. For many people in states with limited abortion access, telehealth remains one of the few practical ways to obtain medication abortion without traveling long distances.
The decision did not end the lawsuit. It only preserved the status quo temporarily.
Justices Samuel Alito and Clarence Thomas dissented, signaling that at least some members of the court remain skeptical of the current rules.
That split made clear the legal battle is far from over.
Mifepristone Is At The Center Of The Fight

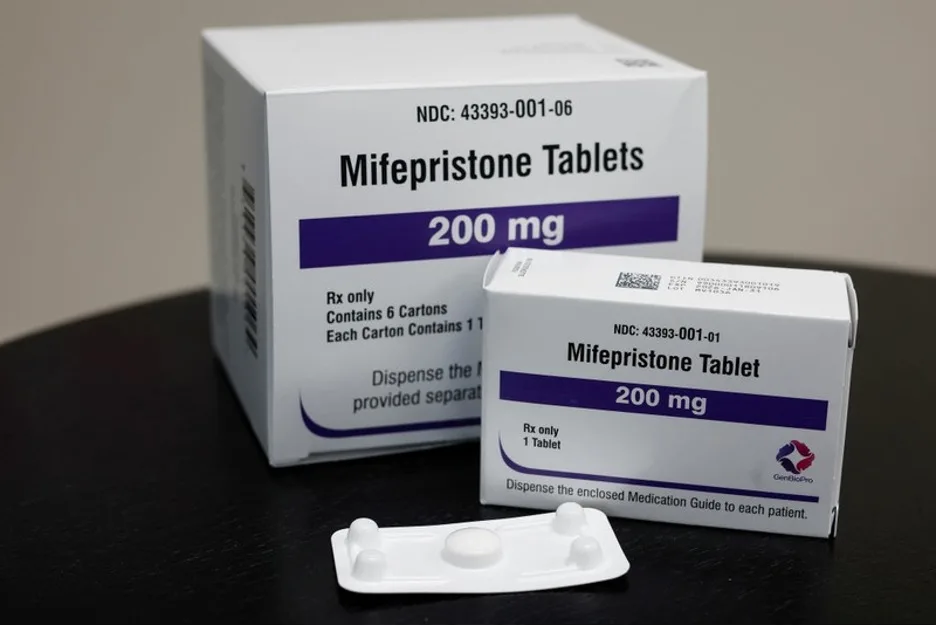
Mifepristone is one of two medications commonly used in the standard medication abortion regimen during the first trimester. It is also used in miscarriage care.
Since the FDA first approved mifepristone in 2000, it has become central to abortion access in the United States. In recent years, telehealth prescribing has made the medication even more important, especially for patients in rural areas or states where clinics are limited.
Abortion-rights advocates argue that mifepristone has a long record of safety and effectiveness. They say restricting telehealth access would create unnecessary barriers for patients and interfere with evidence-based medical care.
Abortion opponents argue that mailing abortion pills without in-person physician oversight places women at risk and undermines state abortion restrictions.
The Supreme Court’s order did not settle that dispute, but it kept the current system operating for now.
Louisiana Challenged The FDA Rule
The case began when Louisiana sued the FDA, arguing that federal rules allowing telehealth access to mifepristone undermine the state’s abortion ban.
Louisiana officials say the FDA’s policy makes it harder for states with strict abortion laws to enforce their own restrictions. They argue that abortion medication can move across state lines through mail and telehealth networks, weakening state-level control.
The 5th Circuit Court of Appeals sided with Louisiana on May 1, creating immediate uncertainty around telehealth abortion access.
Manufacturers Danco Laboratories and GenBioPro then filed emergency appeals, leading the Supreme Court to step in with a temporary stay. That stay was extended until the court’s latest order.
For providers and patients, the back-and-forth created confusion over what was legal, what was available, and how quickly rules might change.
Doctors Warned Confusion Harms Patients

Medical groups welcomed the Supreme Court’s decision but warned that the uncertainty itself is harmful.
Dr. Camille A. Clare, president of the American College of Obstetricians and Gynecologists, said the fight is not over and emphasized that rapid changes to access can harm both patients and clinicians.
Doctors argue that patients need clear, stable medical guidance. When court rulings change access almost overnight, clinics must adjust protocols, pharmacies must interpret legal risk, and patients may delay care while trying to understand their options.
That chaos can be especially difficult for people in states with strict abortion bans, where travel, cost, privacy, and timing already create major barriers.
Medication abortion is time-sensitive. Delays can force patients into more complicated or more expensive care.
Abortion Opponents Condemned The Decision
Anti-abortion groups criticized the Supreme Court’s decision, arguing that states should have more power to restrict medication abortion.
Heartbeat International president Jor-El Godsey said women deserve better than abortion drugs sent by mail without in-person medical support. His statement reflected a broader argument from abortion opponents that telehealth abortion is unsafe and should not override state laws.
Many anti-abortion organizations have pushed the Trump administration to reverse Biden-era rules expanding telehealth access to mifepristone.
They also want the FDA to revisit the drug’s safety review and impose tighter restrictions.
For these groups, the Supreme Court order was not the end of the fight, but another stage in a larger strategy to restrict abortion medication nationally.
The Comstock Act Reentered The Debate

Justice Clarence Thomas raised the Comstock Act in his dissent.
The Comstock Act is an 1873 federal law that banned the mailing of obscene materials and certain items related to abortion. Although the law has long been largely unenforced, some anti-abortion legal advocates have argued it could be used to restrict mailing abortion pills.
During the 2024 presidential campaign, Donald Trump said he did not support using the Comstock Act to stop mail delivery of abortion medication, saying the federal government should not be involved in that issue.
Still, Thomas’ dissent showed that the law remains part of the legal conversation.
That matters because a revival of the Comstock Act could dramatically change abortion access nationwide, even in states where abortion remains legal.
Alito Focused On Irreparable Harm
Justice Samuel Alito also dissented, questioning whether mifepristone manufacturers had shown the kind of irreparable injury necessary to justify relief.
He argued that lost sales in states where abortion-inducing drugs are generally illegal do not automatically qualify as irreparable harm.
His dissent reflected a narrower legal concern, but it carried major practical implications.
If courts become less willing to protect manufacturers from disrupted access rules, medication abortion policy could become more unstable as lawsuits move through different jurisdictions.
That instability could affect not only abortion providers, but also pharmacies, telehealth companies, and patients trying to navigate changing rules.
The FDA Safety Review Adds Another Layer

The lawsuit is also unfolding while the FDA conducts a safety review of mifepristone.
That review was requested after anti-abortion research raised concerns about the drug. Abortion-rights advocates and many medical organizations argue that the drug’s safety record has been well established over decades.
A federal district court had paused the lawsuit at the FDA’s request until that review could be completed, but Louisiana appealed to the 5th Circuit.
The safety review has become politically charged.
Anti-abortion groups accused the FDA of moving too slowly. Some groups publicly praised Acting Commissioner Kyle Diamantas and described him as strongly opposed to abortion.
The leadership change at the FDA has added even more uncertainty to the future of mifepristone access.
Providers Are Preparing Backup Plans
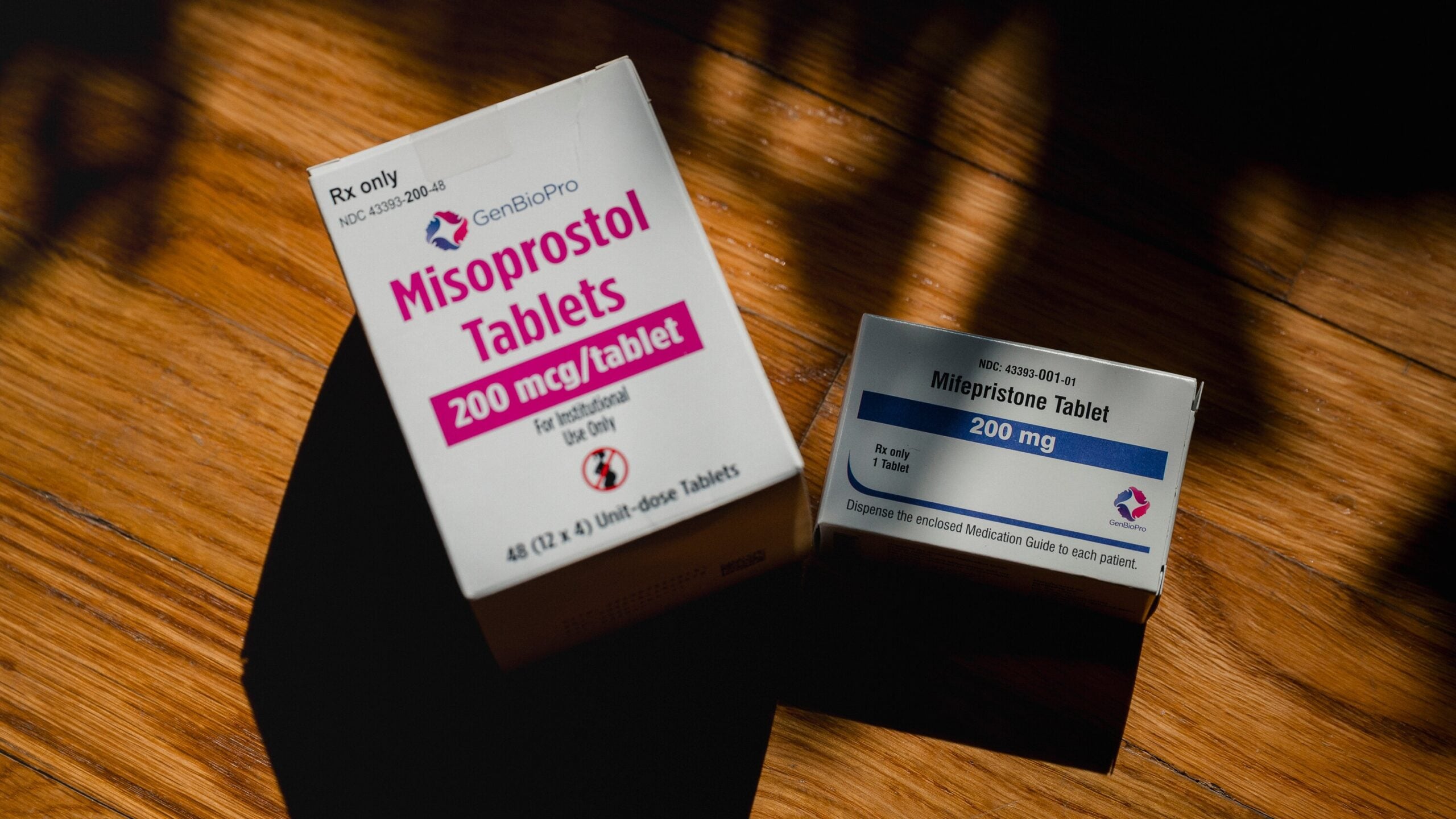
Because of multiple legal threats to mifepristone, abortion providers have been preparing for the possibility that they may need to switch to a misoprostol-only regimen.
Misoprostol can be used alone for medication abortion and miscarriage care. Research has found it to be safe, though it may involve more symptoms and can be slightly less effective than the two-drug regimen using both mifepristone and misoprostol.
Providers say they are preparing these alternatives because legal access to mifepristone remains uncertain.
This preparation shows how deeply court battles are reshaping medical practice.
Doctors are not only making decisions based on clinical evidence. They are also constantly responding to shifting legal risk.
Telehealth Became A Critical Access Point

Telehealth abortion has become especially important since Roe v. Wade was overturned.
In states where abortion is banned or heavily restricted, patients often face long travel distances, high costs, and limited clinic availability. Telemedicine can reduce some of those barriers by allowing patients to consult with providers remotely and receive medication through the mail where legal.
Supporters say this option is essential for people who cannot afford travel, cannot take time off work, or live far from clinics.
Opponents argue telehealth makes it too easy to bypass state abortion laws and reduces medical supervision.
This disagreement sits at the heart of the case.
Doctors Warn Travel Barriers Could Increase
More than 2,200 physicians reportedly signed a letter asking the Supreme Court to maintain telehealth access.
Rob Davidson, an emergency physician in Michigan and executive director of the Committee to Protect Health Care, warned that eliminating telehealth access could force women to travel hundreds of miles for care.
That concern is especially serious for low-income patients, young people, people in abusive relationships, and those living in rural areas.
Travel is not just inconvenient. It can involve transportation costs, childcare, lost wages, hotel stays, and legal uncertainty.
Abortion-rights advocates argue that removing telehealth options would not eliminate demand for abortion medication. It would simply make care harder, later, and more stressful.
The Case Could Shape Abortion Access Nationwide
Although the case began with Louisiana, its impact could reach far beyond one state.
If courts ultimately side with Louisiana and restrict FDA telehealth rules, medication abortion access could change across the country. Even states where abortion remains legal could face confusion if federal regulations are altered.
The case also raises larger questions about whether individual states can challenge FDA drug decisions when they disagree with how a medication is accessed.
Pharmaceutical companies are watching closely because the outcome could affect more than abortion drugs.
If courts allow states to reshape access to federally approved medications based on political objections, drug regulation could become far less predictable.
The Political Battle Is Only Growing
The Supreme Court’s order arrived in the middle of an intense election-year fight over abortion.
Since Roe was overturned, abortion has remained one of the most powerful political issues in the United States. State ballot measures, court decisions, congressional proposals, and federal agency actions have all become part of the same national battle.
The mifepristone case is especially important because medication abortion now accounts for a major share of abortions in the country.
That means any change to telehealth access could affect thousands of patients very quickly.
What Happens Next
For now, telehealth access to mifepristone remains available under current FDA rules while the lawsuit continues.
But the key phrase is “for now.”
The 5th Circuit still has to rule on the merits of the case. The FDA safety review remains politically charged. Anti-abortion groups are continuing to pressure federal officials. Abortion-rights advocates are preparing for more court fights.
The Supreme Court’s decision brought temporary stability, but not final certainty.
For patients and doctors, that uncertainty has become the defining feature of abortion access in the United States.
The larger question remains unresolved: will medication abortion be governed primarily by federal medical regulators, state abortion bans, old federal laws, or shifting court orders?
Until that question is settled, access to mifepristone may continue to change not through medical evidence alone, but through lawsuits, politics, and emergency court decisions that can reshape healthcare overnight.