Table of Contents
- What Happened? The FDA’s Withdrawal of Vaccine Studies
- The Trump Administration’s Stance on Vaccines
- A Deeper Look: What Were the Studies About?
- The Fallout: How Will This Impact Public Perception?
- The Role of the FDA in Public Health Policy
- Vaccine Safety and Public Trust
- What Does This Mean for the Future of Vaccines?
- The Political Context: Why Is This Happening?
- Legal and Ethical Concerns
- The Need for Stronger Public Health Messaging
- A Turning Point for Vaccine Policy
What Happened? The FDA’s Withdrawal of Vaccine Studies
The FDA’s decision to block the publication of multiple studies on Covid and shingles vaccines has become a hot topic in the ongoing debate over vaccine safety. The studies, which were based on millions of patient records, aimed to assess the safety profiles of vaccines, particularly focusing on side effects. However, the studies were withdrawn when it was discovered that their authors had drawn conclusions that were deemed too broad given the underlying data. A spokesperson from the U.S. Department of Health and Human Services (HHS) confirmed that the studies did not meet the FDA’s “high standards” and were pulled to protect the integrity of the scientific process.
The Trump Administration’s Stance on Vaccines

This move comes as part of a broader trend under the Trump administration to challenge the safety and accessibility of vaccines in the United States. Health and Human Services Secretary Robert F. Kennedy Jr., a prominent vaccine skeptic, has been at the forefront of efforts to revise Covid shot recommendations, limit research on vaccine development, and even overhaul the childhood immunization schedule. His controversial stance on vaccines has prompted concern among public health experts, who argue that these actions could undermine trust in essential vaccines and make it more difficult for some Americans to access life-saving shots.
A Deeper Look: What Were the Studies About?
The studies that were withdrawn by the FDA were comprehensive, utilizing data from millions of patient records to assess the prevalence of side effects related to the Covid and shingles vaccines. The findings suggested that side effects from the vaccines were rare, and that the benefits far outweighed the risks for most patients. These studies were expected to provide reassurance to the public and bolster confidence in vaccine safety. However, the FDA decided to block their publication after reviewing the conclusions drawn by the authors, arguing that the data did not fully support the broad claims made in the studies.
The Fallout: How Will This Impact Public Perception?
The FDA’s decision to withdraw these studies has sparked backlash, particularly among those who believe that the Trump administration is deliberately undermining public confidence in vaccines. Many experts believe that such actions could further fuel skepticism surrounding the safety and effectiveness of vaccines, especially as the world continues to grapple with the Covid-19 pandemic and its aftermath. With vaccine hesitancy already on the rise, particularly in certain political and social groups, the withdrawal of these studies is seen as a significant blow to efforts to reassure the public about the safety of vaccines.
The Role of the FDA in Public Health Policy
The FDA’s role in regulating and ensuring the safety of vaccines is crucial, and its actions can have far-reaching consequences for public health. The agency is responsible for approving vaccines, overseeing clinical trials, and monitoring their safety once they are on the market. Its decision to block the publication of studies supporting the safety of vaccines raises questions about its independence and the extent to which political pressures are influencing its decision-making. Health experts are now calling for greater transparency and accountability from the FDA to ensure that the public can trust the agency’s actions and recommendations.
Vaccine Safety and Public Trust
Vaccine safety is one of the most critical public health issues of our time. For decades, vaccines have been instrumental in preventing the spread of infectious diseases and saving millions of lives worldwide. However, the rise of vaccine misinformation and skepticism has led to a decline in vaccination rates, leaving individuals and communities vulnerable to preventable diseases. In this context, the FDA’s withdrawal of vaccine safety studies is deeply concerning, as it may further erode public trust in vaccines and the agencies responsible for their oversight.
What Does This Mean for the Future of Vaccines?
The ongoing controversy surrounding the FDA’s actions could have significant implications for the future of vaccines in the United States. As vaccine hesitancy continues to grow, it is essential that health authorities take proactive steps to rebuild trust in the safety and effectiveness of vaccines. This may involve increased transparency in the approval and oversight process, as well as a commitment to unbiased, evidence-based decision-making. If public confidence in vaccines continues to decline, the consequences could be dire, leading to the resurgence of diseases that were previously under control.
The Political Context: Why Is This Happening?
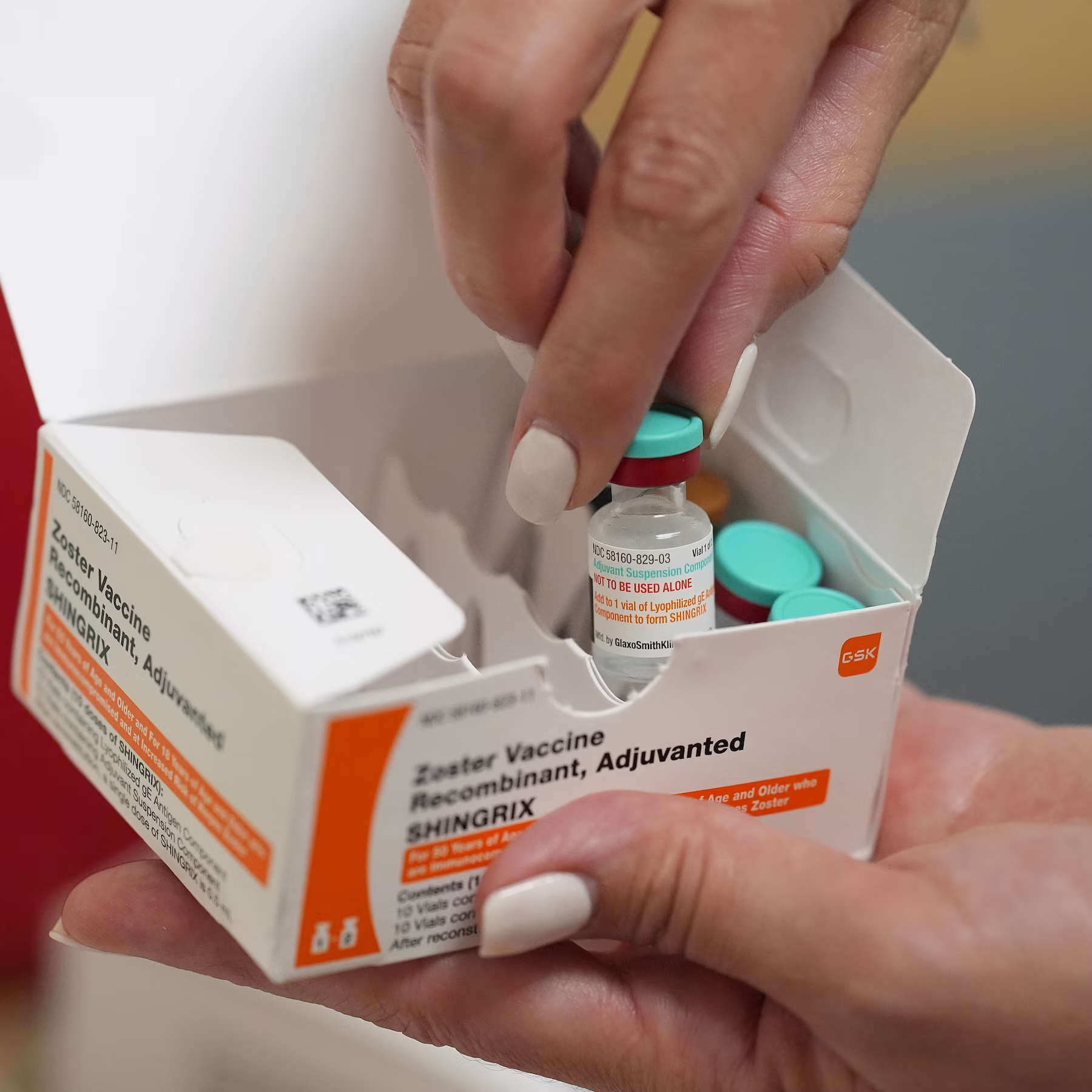
The political context surrounding the FDA’s withdrawal of vaccine safety studies is also important to understand. Under the Trump administration, there has been a concerted effort to challenge vaccine mandates and loosen regulations on vaccine development. This aligns with the broader trend of skepticism toward government health policies that has been prevalent in certain political circles. With figures like Robert F. Kennedy Jr. at the helm of HHS, there is a growing movement to roll back public health measures that are seen as overly restrictive. This political climate has created an environment in which the safety of vaccines is increasingly questioned, even as the science continues to support their effectiveness.
Legal and Ethical Concerns
The decision to block the publication of these studies raises several legal and ethical concerns. First, there is the issue of transparency: If studies on vaccine safety are being blocked or altered, it undermines the scientific process and limits the public’s access to critical health information. Additionally, there are concerns about the potential influence of political pressures on scientific research. The FDA’s decision to withdraw these studies could set a dangerous precedent, where political considerations outweigh scientific evidence and public health priorities.
The Need for Stronger Public Health Messaging

In the wake of this controversy, there is an urgent need for stronger public health messaging and communication from the FDA and other health agencies. Clear, consistent information about the safety and efficacy of vaccines is essential to combat misinformation and build trust in public health systems. This includes being transparent about the research process, addressing concerns about vaccine side effects, and providing the public with reliable sources of information. Only by taking these steps can health authorities hope to restore faith in vaccines and ensure that future outbreaks are prevented.
A Turning Point for Vaccine Policy
The FDA’s decision to block studies supporting the safety of Covid and shingles vaccines is a significant moment in the ongoing debate over vaccine safety and public health policy in the United States. While the withdrawal of these studies may be framed as a move to protect scientific integrity, it raises important questions about the influence of political pressures on public health decisions. The fallout from this controversy is likely to have far-reaching consequences for the future of vaccines in the U.S. and the global efforts to combat infectious diseases.