Table of Contents
- Why Chronic Low Back Pain Has Become Such a Massive Global Problem
- The Limits of Conventional Treatments
- A New Class of Injectable Disc Treatments Is Taking Shape
- The Hydrogel Approach Designed to Fill, Seal, and Rehydrate the Disc
- Early Results That Have Generated Real Excitement
- A Protein-Based Biologic That Tries to Restore Disc Biology
- Why Pain Signaling Matters as Much as Structural Repair
- A Cell Therapy Built to Rebuild the Disc Matrix
- Why This Field Could Change the Future of Spine Care
- What Happens Next for Patients and the Industry
Why Chronic Low Back Pain Has Become Such a Massive Global Problem
Low back pain remains one of the most common and disabling health problems in the world. It affects work, sleep, exercise, mental health, and overall quality of life. For some people it comes and goes, but for others it becomes a daily burden that shapes how they sit, walk, bend, drive, and even socialize. The frustration is not just the pain itself. It is the feeling that the condition often lingers for years, with treatments that manage discomfort without fully addressing the source.
In many cases, that source is degeneration of the intervertebral disc. These discs sit between the vertebrae and act like cushions, helping the spine absorb shock and maintain flexibility. At the center is the nucleus pulposus, a gel-like core that contains water and collagen-rich material. Around it sits the annulus fibrosis, a tougher outer ring that keeps the disc contained. When degeneration starts, the disc can lose hydration, weaken structurally, and become less capable of handling normal loads. Over time, that breakdown can trigger inflammation, instability, nerve irritation, and chronic pain.
The Limits of Conventional Treatments
For years, standard treatment for degenerative disc disease has relied on anti-inflammatory medications, physical therapy, activity modification, weight management, and general lifestyle improvements. These methods can be helpful, and for many patients they remain essential first steps. They may reduce flare-ups, improve mobility, and help people function better. Still, they often do not reverse the damage inside the disc.
That gap in care is exactly where researchers see opportunity. Instead of waiting until degeneration becomes severe enough to justify surgery, scientists and biotech companies are working on injectable therapies designed to intervene earlier. The idea is both practical and ambitious. Deliver a treatment directly into the damaged disc, use the disc’s own space as the target, and try to stabilize or regenerate the tissue before the problem worsens. It is a concept that sounds simple in theory, but the science behind it is highly sophisticated.
A New Class of Injectable Disc Treatments Is Taking Shape
Several research groups are now testing different ways to treat damaged spinal discs from within. Although their technologies differ, they all share the same broad goal: reduce pain, restore function, and preserve as much of the natural disc as possible. Among the most closely watched approaches are a hydrogel implant, a protein-based biologic, and a disc cell therapy using differentiated progenitor cells.
What makes this moment especially notable is that these treatments are not all chasing the exact same patient. Some are being designed for mild to moderate degeneration, while others may be more suitable for moderate to severe disease. That suggests the future of spinal care may become more tailored, with doctors choosing among biologics, implants, or cell therapies depending on the stage of disc failure and the patient’s symptoms.
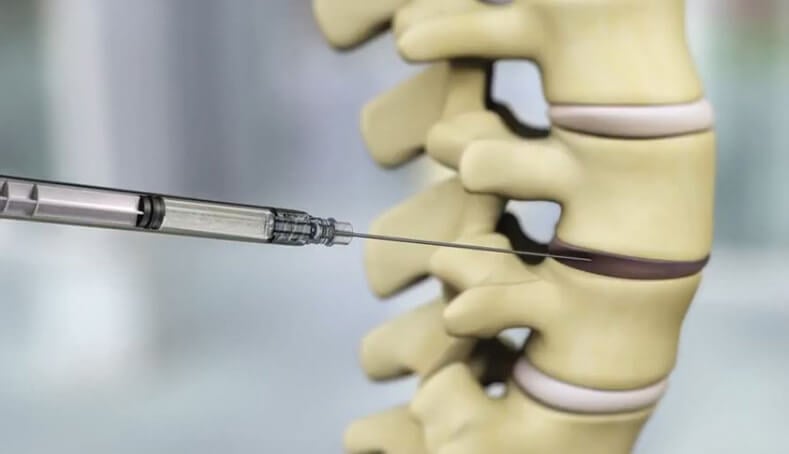
The Hydrogel Approach Designed to Fill, Seal, and Rehydrate the Disc
One of the most intriguing options under development is HYDRAFIL, a gel created by ReGelTec. Unlike a traditional implant that fully replaces disc material, this injectable hydrogel is meant to augment what remains. It is designed to fill the nucleus pulposus space and also help seal tears in the annulus fibrosis, which can become especially significant in more advanced degeneration.
The concept behind HYDRAFIL is appealing because it attempts to do several jobs at once. First, it restores some of the mechanical loading capacity of the disc, which can help the spine function more normally. Second, its hydrophilic properties allow it to attract and retain water, which is crucial because a healthy disc depends on hydration. Third, because it is delivered in liquid form and solidifies in place, it can be inserted through a small needle rather than through major surgery.
That liquid-to-solid transition was a major breakthrough in the product’s development. Researchers modified earlier gel designs and focused on polymer interactions, thermodynamics, and processing so the material could be heated into an injectable liquid, then cool into a stable gel once inside the disc. It is the kind of engineering challenge that blends biomaterials science with practical medicine. The end result is a treatment intended not to make the disc perfect again, but to make it more functional and less painful.
Early Results That Have Generated Real Excitement
Preclinical testing helped establish feasibility, and clinical work that followed offered encouraging signs. In early patient treatment, HYDRAFIL was injected into affected discs under fluoroscopic guidance. According to reported outcomes, average pain scores dropped dramatically over the following months. Later follow-up data from additional patients in Colombia and Canada suggested that this pain reduction was not just immediate but potentially durable, with some patients maintaining strong improvement out to one and even two years.
Researchers also tracked disability scores, not just pain ratings, which matters because chronic back pain is not only about discomfort. It is about what pain takes away. In these early findings, patients moved from severe disability ranges before treatment to mild disability ranges after treatment. That kind of shift is exactly what makes clinicians pay attention. It suggests the therapy may be doing more than masking symptoms.
Another advantage is that the gel does not appear to burn future treatment options. If a patient later needs another procedure, the material can reportedly be removed with a relatively simple endoscopic technique. That flexibility could become a meaningful selling point in a field where physicians are often cautious about any therapy that might complicate future care.
A Protein-Based Biologic That Tries to Restore Disc Biology
A very different strategy is being pursued by Notogen through its protein-based biologic NTG-101. This therapy is rooted in years of research into notochordal cells, which appear to have a protective influence on disc health. Scientists investigating certain dog breeds that are more resistant to disc degeneration found clues suggesting that proteins secreted by these cells may help preserve or restore the disc environment.
Over time, researchers narrowed their focus to two proteins, transforming growth factor beta 1 and connective tissue growth factor. Together, these became the foundation of NTG-101. Instead of acting like a structural filler, this therapy aims to influence the biology of the disc itself. In simple terms, it tries to encourage the disc toward repair by promoting beneficial matrix production and suppressing destructive processes associated with degeneration.
In animal models, including canine studies, the results were striking enough to attract growing attention. Injured discs treated with NTG-101 showed features that looked far closer to normal discs than untreated controls. Researchers reported increased proteoglycans and collagen type II, both of which are essential for disc health. At the same time, they saw suppression of the catabolic enzymes that tend to drive degeneration forward.
Why Pain Signaling Matters as Much as Structural Repair
One of the most interesting aspects of the NTG-101 research is that it extended beyond structural repair. Scientists also looked at markers associated with pain, including neurotrophins and neuropeptides known to rise in painful disc conditions. In untreated injured discs, those pain-related signals increased sharply. In discs treated with NTG-101, they remained closer to normal levels.
That matters because a disc can look better on imaging without necessarily solving the patient’s pain. Chronic pain is shaped by both structure and biology. A therapy that influences inflammation, cellular signaling, and tissue composition all at once may have a better chance of producing meaningful relief. Notogen’s work remains earlier on the path to commercialization, but its scientific rationale is clearly one of the reasons it continues to generate interest.
A Cell Therapy Built to Rebuild the Disc Matrix
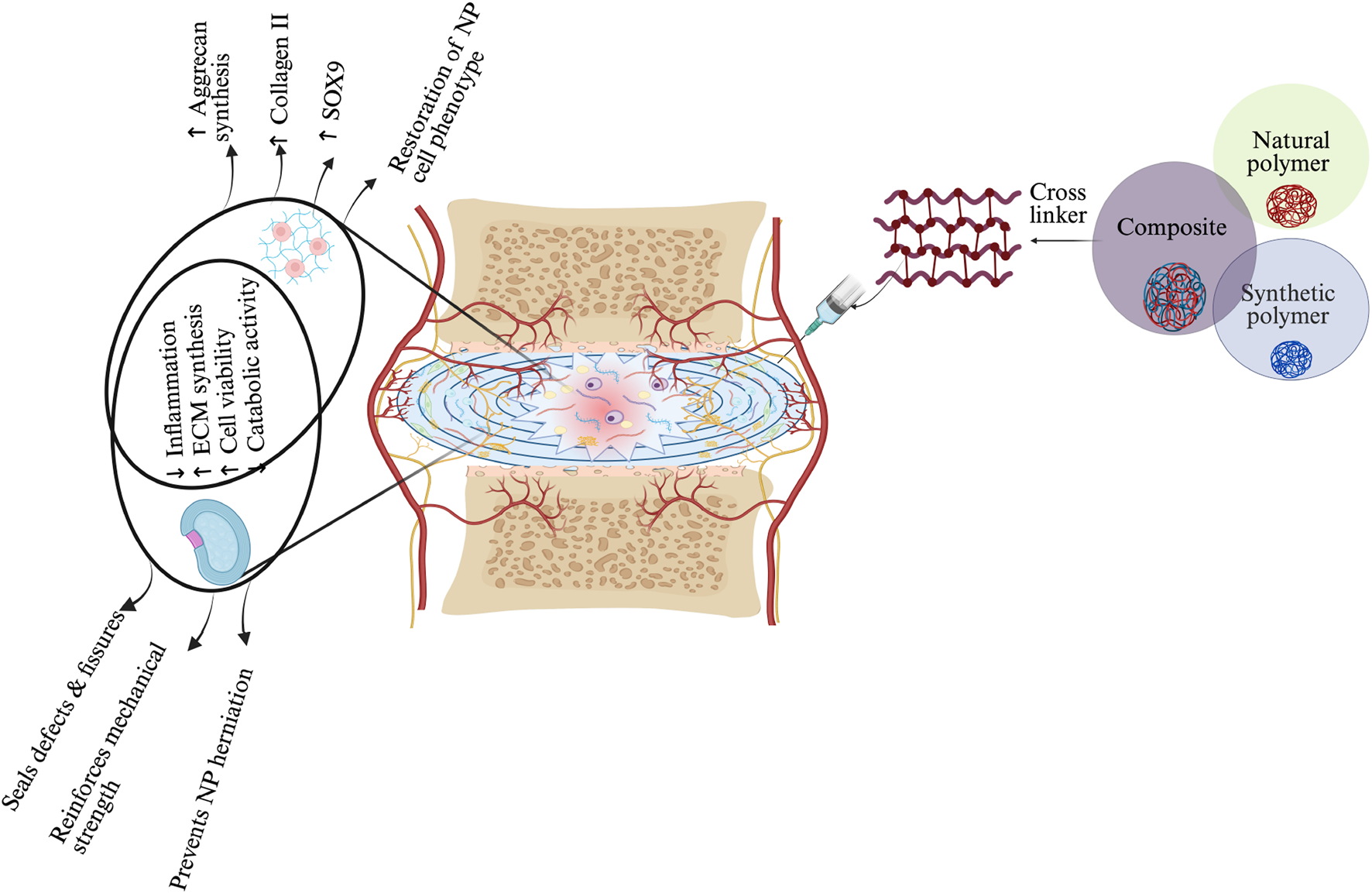
DiscGenics is taking yet another route with its injectable disc cell therapy, known as IDCT. This product combines millions of differentiated progenitor cells, called discogenic cells, with a proprietary viscous carrier that helps protect the cells and distribute them throughout the disc. The logic behind this method is that the cells are already specialized for disc repair, making them potentially more durable and regenerative than more generalized stem cell approaches.
The importance of proteoglycans again comes into focus here. These molecules help discs hold water, which keeps them cushioned and functional. As degeneration progresses, proteoglycans disappear, disc hydration falls, and the disc becomes more vulnerable to collapse and instability. By introducing discogenic cells that are already producing disc matrix components such as collagen and proteoglycans, IDCT is designed to help rebuild what degeneration has stripped away.
According to the company, studies in animals and humans have shown encouraging results. In preclinical models, disc height improved within weeks. In human trials, patients showed meaningful gains in pain scores, disability measures, quality of life, and even disc volume. High-dose IDCT results have reportedly remained durable through two years, which is the kind of timeline that investors, regulators, and patients all want to see.
Why This Field Could Change the Future of Spine Care
Taken together, these technologies represent something bigger than three isolated products. They suggest that the treatment of degenerative disc disease may be entering a new era. For a long time, patients with chronic back pain have faced a frustrating gap between conservative care and surgery. Injectable disc treatments may eventually help fill that gap with options that are less invasive yet more biologically meaningful than pain management alone.
That does not mean every patient will be a candidate, or that every therapy will reach the market on schedule. Clinical trials can disappoint. Regulators can raise tough questions. Manufacturing complex biologics and cell therapies is expensive and technically demanding. Long-term durability must still be proven. Even so, the direction of the science is hard to ignore. Researchers are no longer asking only how to calm the pain. They are asking how to restore disc health itself.
What Happens Next for Patients and the Industry

The next few years will be crucial. ReGelTec has moved into larger FDA-directed study plans. DiscGenics has been preparing late-stage clinical work and manufacturing scale-up. Notogen continues building the case for its molecular therapy with the help of collaborators and industry support. Each company is on a different timeline, but all are moving toward the same question: can these injectable disc treatments deliver safe, lasting, clinically meaningful relief in real patients at scale?
That question matters far beyond the lab. If even one of these approaches succeeds in a major way, it could reshape how doctors think about chronic back pain. Instead of telling patients to manage symptoms until the disc deteriorates further, clinicians may someday be able to intervene earlier with targeted therapies that protect function and delay more invasive procedures. That would not just be a scientific advance. It would be a practical, deeply human one.
For the millions of people whose lives have been narrowed by degenerative disc disease, that possibility is what makes this story so powerful. Chronic back pain has long been treated as something to endure, control, or work around. These injectable disc treatments suggest a different future, one in which the damaged disc itself becomes the focus of repair. If ongoing trials continue to deliver promising results, this field may not simply ease pain. It may redefine what recovery looks like for one of the world’s most common disabling conditions.